一. Bischler–Napieralski reaction
The ring closure and dehydration of N-acyl β-phenylethylamine in dehydrating reagents (POCl3, P2O5, PPA, TFAA, or Tf2O) to give 3,4-dihydroisoquinoline is called Bischler–Napieralski reaction. This reaction was first reported by Bischler and Napieralski in 1893 [Bischler, A.; Napieralski, B. Ber. 1893, 26, 1903–1908]. This reaction is equivalent to the intramolecular Friedel-Crafts acylation reaction. When POCl3 is used as a dehydrating agent, the mechanism is similar to the intramolecular Vilsmeier-Haack formylation reaction, so the electron-rich aromatic ring can get a higher yield. When the α-position of the substrate amine is substituted with a hydroxyl group, an extra molecule of water will be removed to obtain isoquinoline. The dihydroisoquinoline generated by the reaction can also be converted into isoquinoline by oxidation. The reaction is usually refluxed in toluene or xylene, and there are also reports of reactions in low-boiling acetonitrile or dichloromethane.

二. Gabriel–Colman rearrangement reaction Rearrangement of aryl phthalimidoacetates under basic conditions to give 1,4-diphenolisoquinoline analogues.

三. Synthesis of Pictet–Gams isoquinoline The acyl derivative of β-hydroxy-β-phenylethylamine is refluxed in an inert reagent such as decahydronaphthalene in the presence of a dehydrating reagent (such as phosphorus pentoxide or phosphorus oxychloride) to obtain isoquinoline compounds.

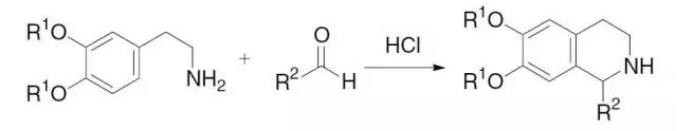
四. Synthesis of Pictet–Spengler Tetrahydroisoquinoline Cyclocondensation of β-arylethylamine and carbonyl compound to obtain tetrahydroisoquinoline. The reaction was reported by Swiss chemists Amé Pictet and Theodor Spengler in 1911. It has a history of nearly 100 years. Now this reaction is still the most effective method for synthesizing tetrahydroisoquinoline and β-carboline derivatives. It is often used in isoquinoline In the synthesis of quinoline alkaloids and drugs.
五. Pomeranz–Fritsch reaction The reaction of aminoacetal and benzaldehyde under acid catalysis to obtain isoquinoline and its saturated derivatives. This reaction is named after Paul Fritsch (1859–1913) and Cäsar Pomeranz (1860–1926). The classic reaction conditions are catalyzed by sulfuric acid or HCl, and Lewis acids (such as BF3, TiCl4, or trifluorosulfonates of lanthanide metals, etc.) and trifluoroacetic anhydride can also catalyze this reaction. Improved Schlittler-Muller method The reaction of phenethylamine and aldehyde acetal condensation and ring closure under acidic conditions to obtain isoquinoline is similar to the Pomeranz–Fritsch reaction, except that the reaction substrate is reversed.

六. Reissert reaction A three-component condensation of quinoline or isoquinoline in the presence of an acid halide and a cyano source gives the Reissert compound, followed by hydrolysis to give the aldehyde, the reaction of quinolinecarboxylic acid and ammonia gas. A variant of this reaction that uses N-oxides instead is known as the Reissert-Henze reaction.

七. Alper carbonylation reaction Halogenated aromatic hydrocarbons containing aliphatic amines, alkenyl amines, alkenyl aziridines, etc., and carbon monoxide are catalyzed by Pd, Ru, or Rh, etc., to prepare cyclic amides by carbonyl ring closure.


